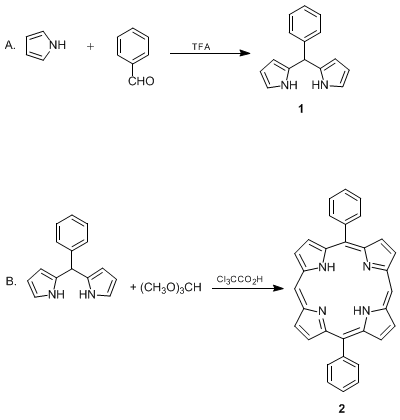
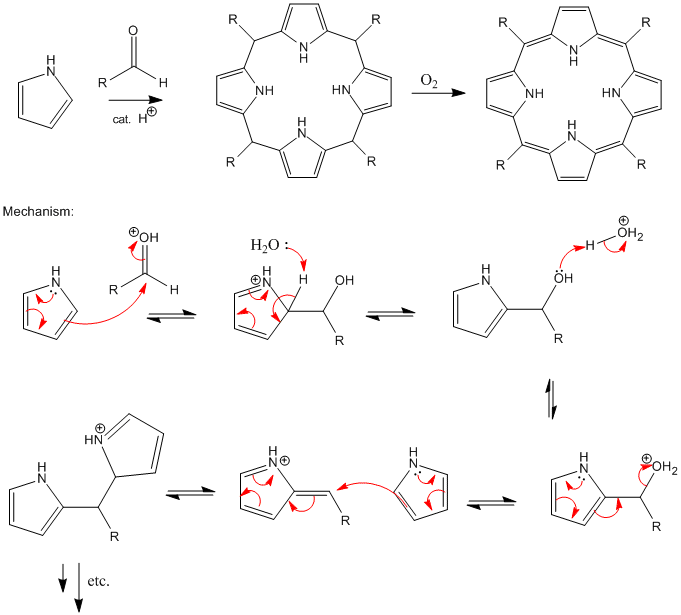
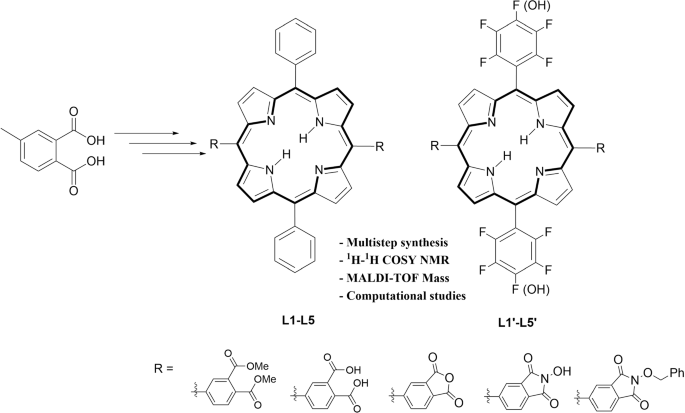
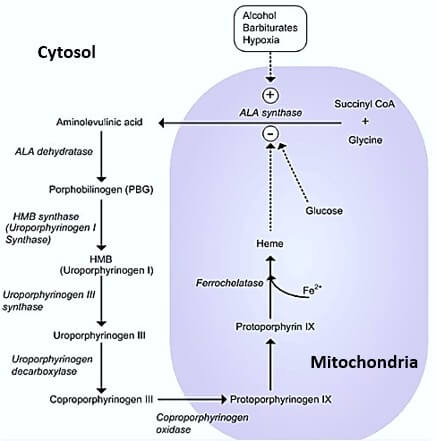
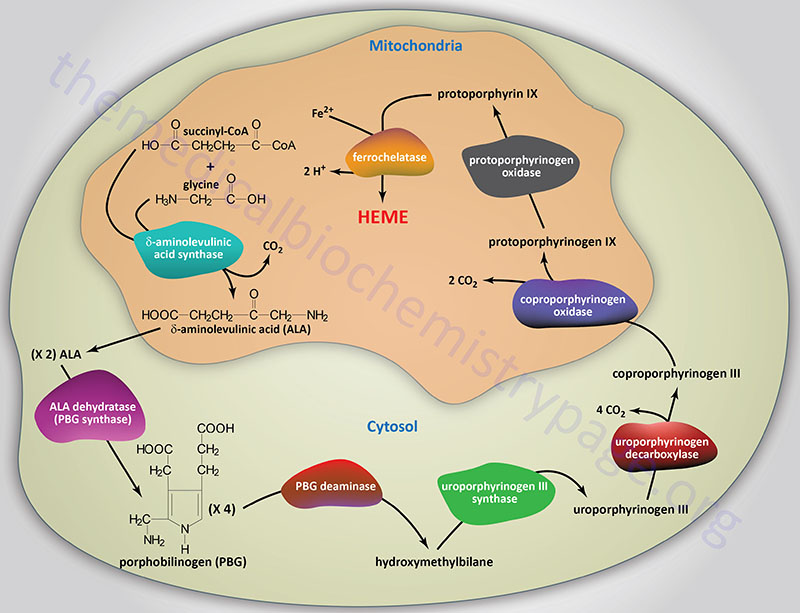
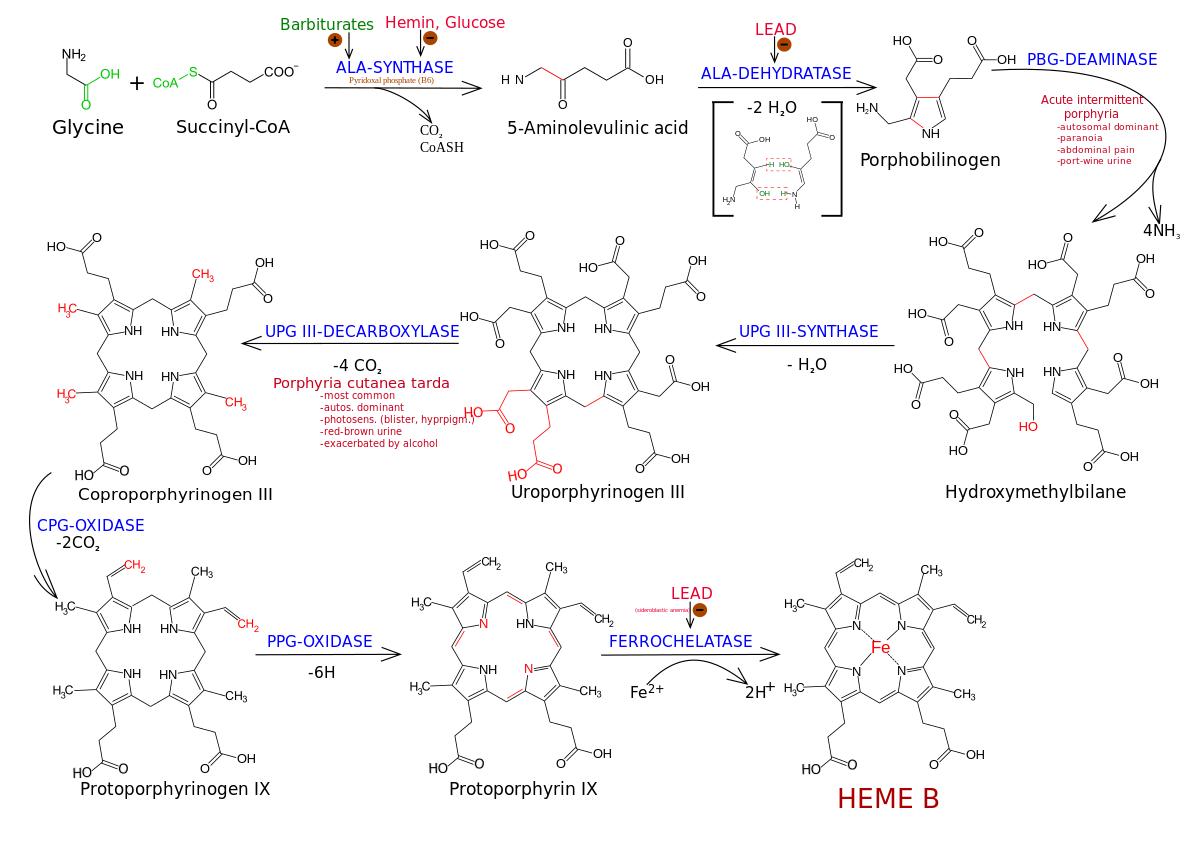
Synthesis and Functionalization of Porphyrins through Organometallic Methodologies | Chemical Reviews
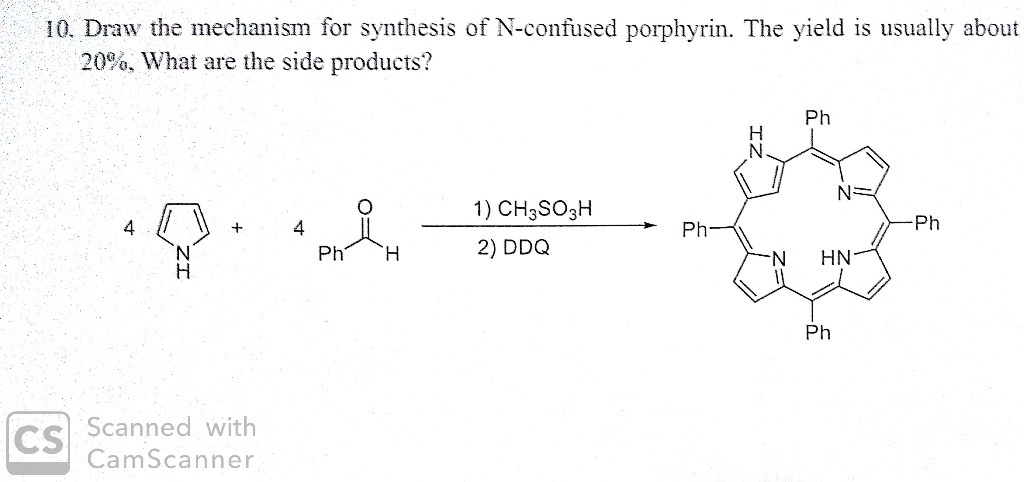
SOLVED:10. Draw the mechanism for synthesis of N-confused porphyrin. The yield is usually about 2096. What are the side products? Ph 1) CHaSOzH 2) DDQ Ph- Ph Ph HN- Ph CS Scanned
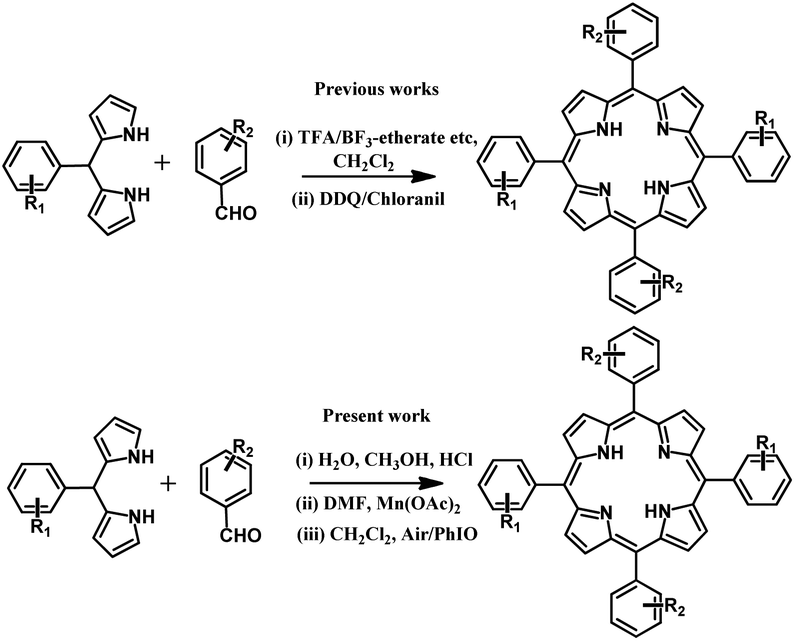
A new synthesis of porphyrins via a putative trans -manganese( iv )-dihydroxide intermediate - Dalton Transactions (RSC Publishing) DOI:10.1039/C9DT03573G

Volume # 1(104), January - February 2016 — "Facile preparation of pure 5,10,15,20-tetrakis(perfluoroalkyl)porphyrins"

Synthesis and characterization of free base and metal porphyrins and their interaction with CdTe QDs - ScienceDirect

Acid Activation and Chemical Oxidation in the Synthesis of meso‐ Tetraphenylporphyrin using a Mixed‐Solvent System - Sun - 2019 - Asian Journal of Organic Chemistry - Wiley Online Library

Synthesis and Functionalization of Porphyrins through Organometallic Methodologies | Chemical Reviews