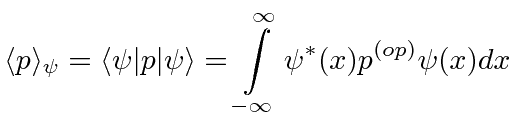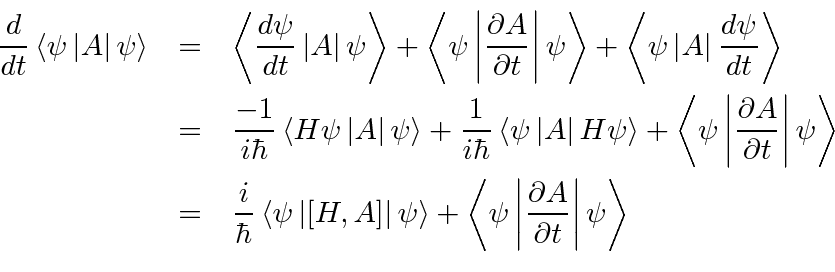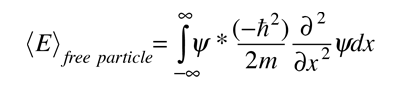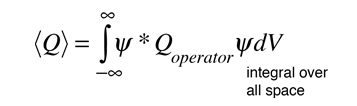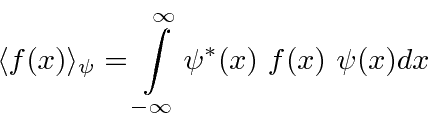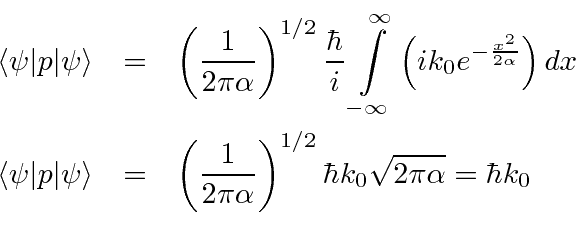Physics - Ch 66 Ch 4 Quantum Mechanics: Schrodinger Eqn (29 of 92) Expect. Value Momentum=? 1-D Box - YouTube

Expectation value of position. Plot of the mean value of position for... | Download Scientific Diagram

CHEM6085: Density Functional Theory Lecture 2. Hamiltonian operators for molecules - PDF Free Download

SOLVED:In quantum mechanics, observables are represented by operators. The operator G(x) corresponds to the observable G(x). Furthermore, we can only determine the expectation value (mean value) of an observable, such as <G(x)> .

The Quantum Theory of Atoms and Molecules The Schrödinger equation and how to use wavefunctions Dr Grant Ritchie. - ppt download