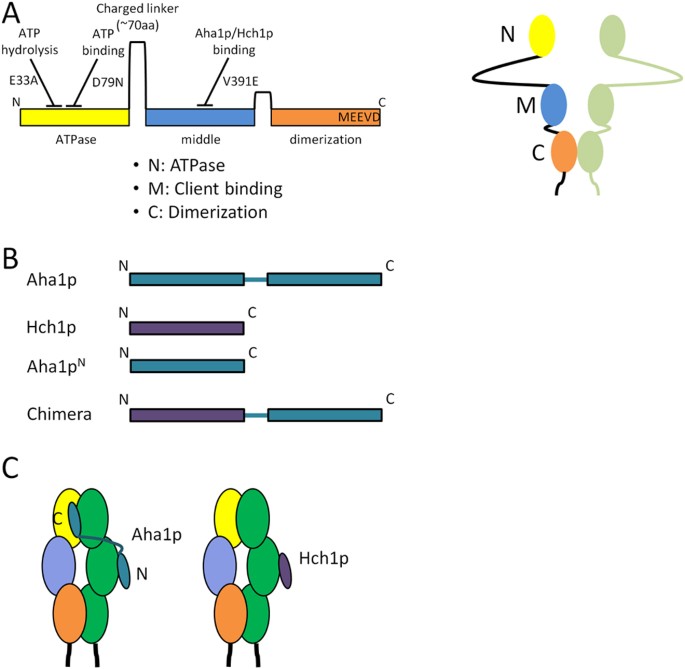
Structural Asymmetry in the Closed State of Mitochondrial Hsp90 (TRAP1) Supports a Two-Step ATP Hydrolysis Mechanism - ScienceDirect

HSP90 and co‐chaperones: a multitaskers' view on plant hormone biology - Donato - 2019 - FEBS Letters - Wiley Online Library

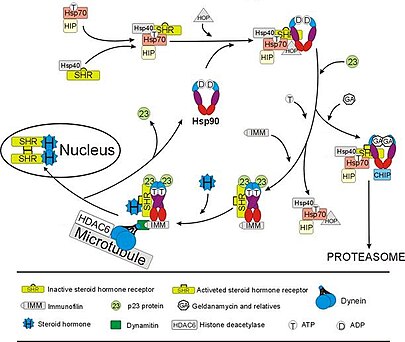
Hsp90 epigenetic mechanism of action. Hsp90 epigenetically regulates... | Download Scientific Diagram

Post-translational modifications of Hsp90 and translating the chaperone code - Journal of Biological Chemistry

Molecular Mechanism of Protein Kinase Recognition and Sorting by the Hsp90 Kinome-Specific Cochaperone Cdc37: Molecular Cell
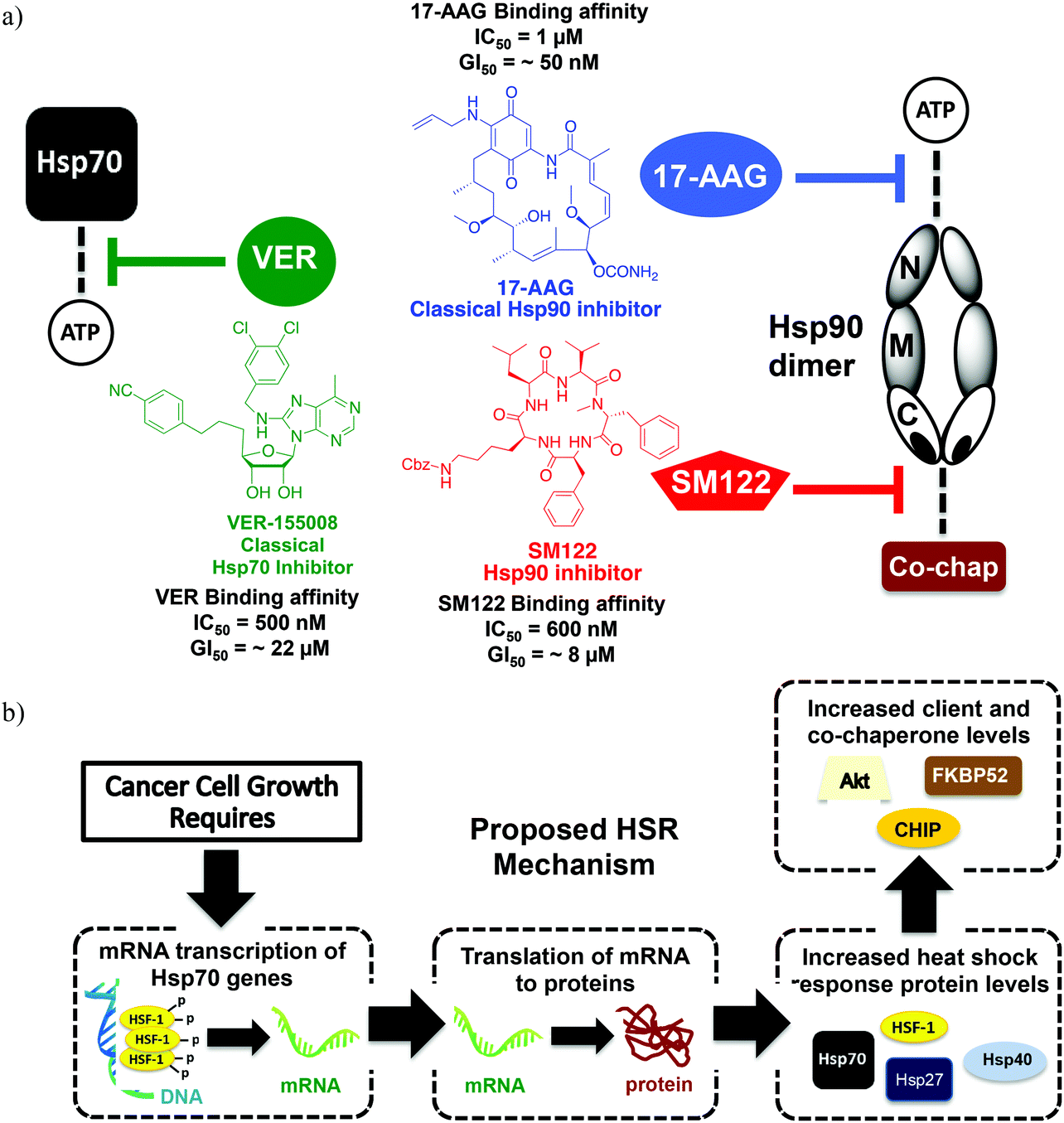
Combining an Hsp70 inhibitor with either an N- or C-terminal Hsp90 inhibitor produces mechanistically distinct phenotypes - Organic & Biomolecular Chemistry (RSC Publishing) DOI:10.1039/C5OB00147A

GR chaperone cycle mechanism revealed by cryo-EM: reactivation of GR by the GR:Hsp90:p23 client-maturation complex | bioRxiv
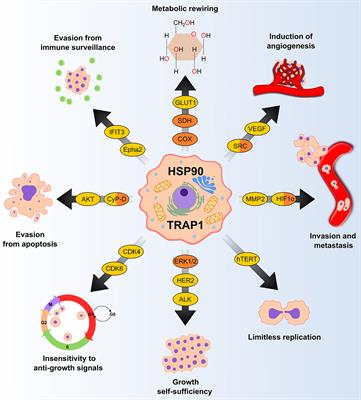
Frontiers | Dynamically Shaping Chaperones. Allosteric Modulators of HSP90 Family as Regulatory Tools of Cell Metabolism in Neoplastic Progression | Oncology
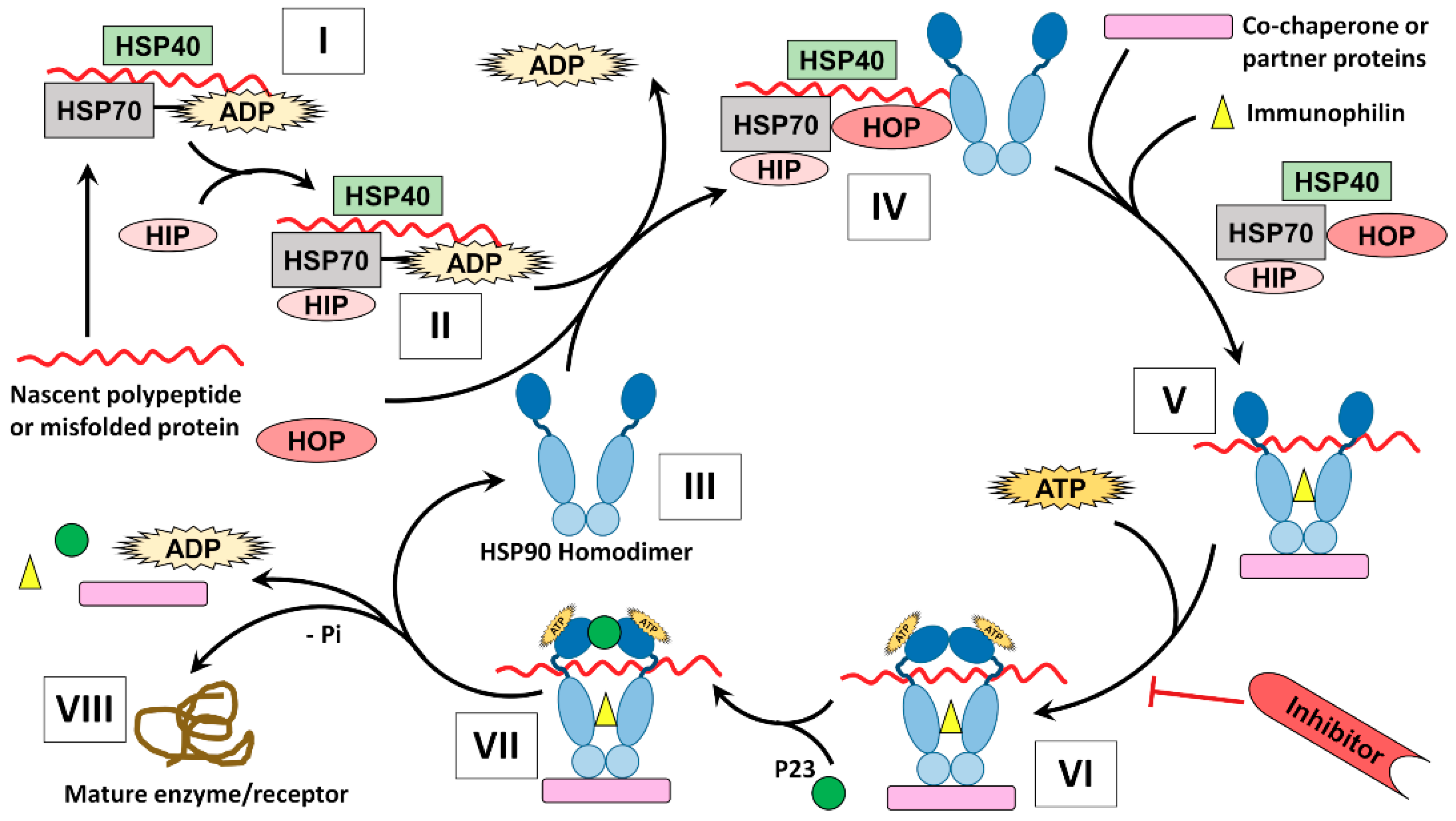
IJMS | Free Full-Text | The HSP90 Family: Structure, Regulation, Function, and Implications in Health and Disease | HTML

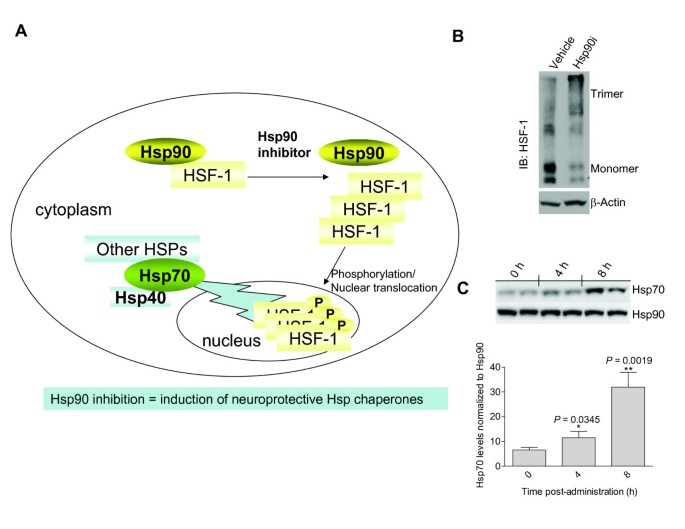
JCI - The high-affinity HSP90-CHIP complex recognizes and selectively degrades phosphorylated tau client proteins